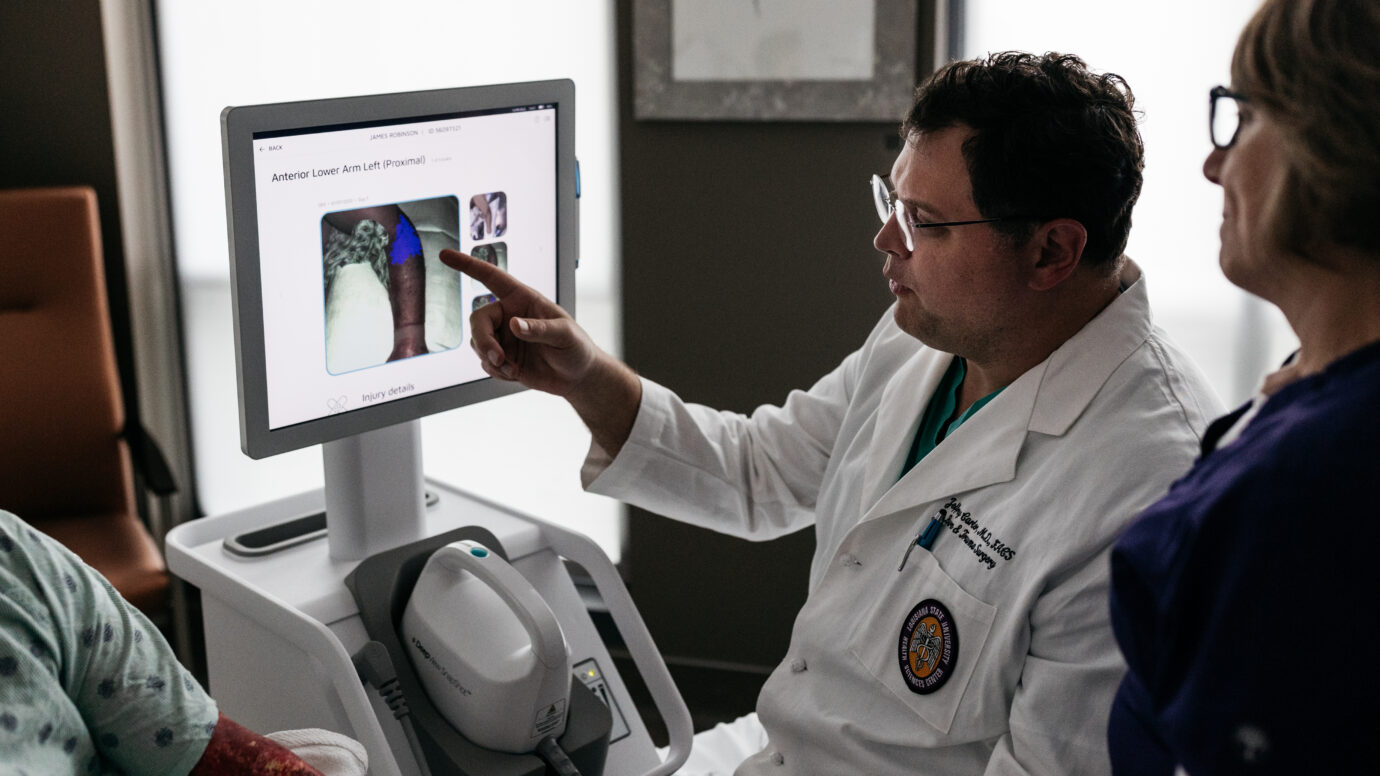
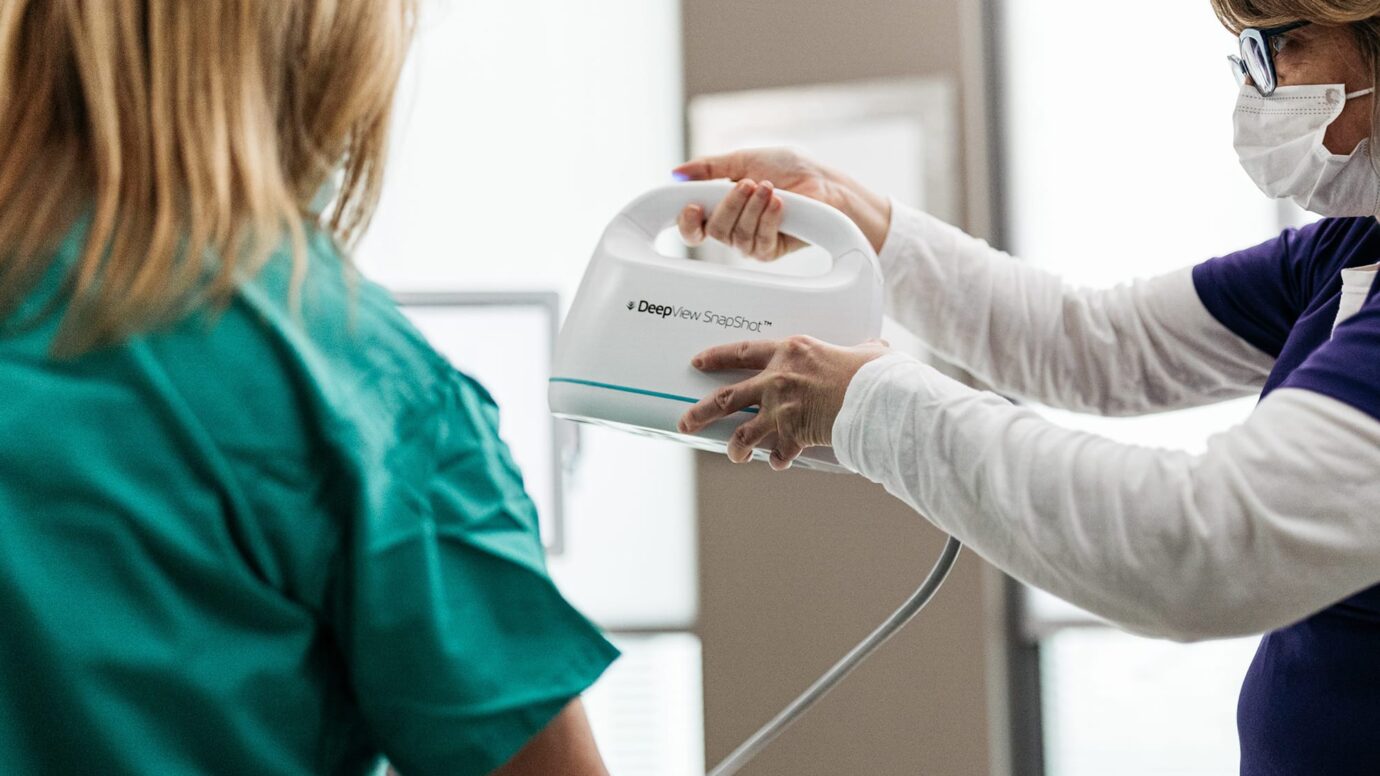
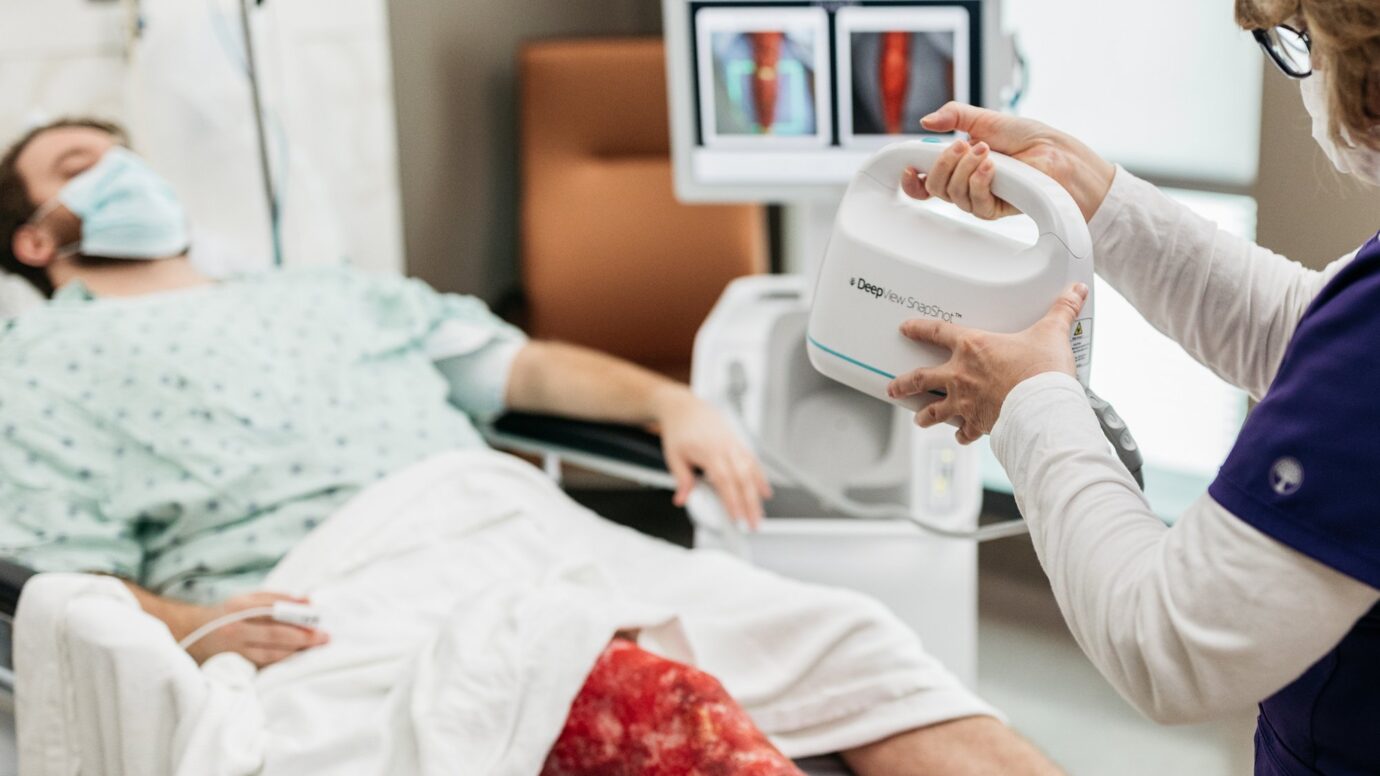
The DeepView® Wound Imaging System
Technology that provides an immediate wound healing prediction to clinicians with information that is not visible to the human eye, using multispectral imaging and Artificial Intelligence.
Noninvasive diagnostic technology using multispectral imaging to improve outcomes for patients with acute and chronic wounds.
Medical imaging with a platform that uses proprietary Artificial Intelligence (AI) to predict how a wound will heal.
Day One immediate binary output for clinicians that allows for a clear healing map of the wound prior to treatment or other interventions.

%
Accurate in predicting if a burn wound will heal or not.
%
Accurate in predicting if a DFU will close 50% of area by week 4.
10 US patents and 10 foreign patents